 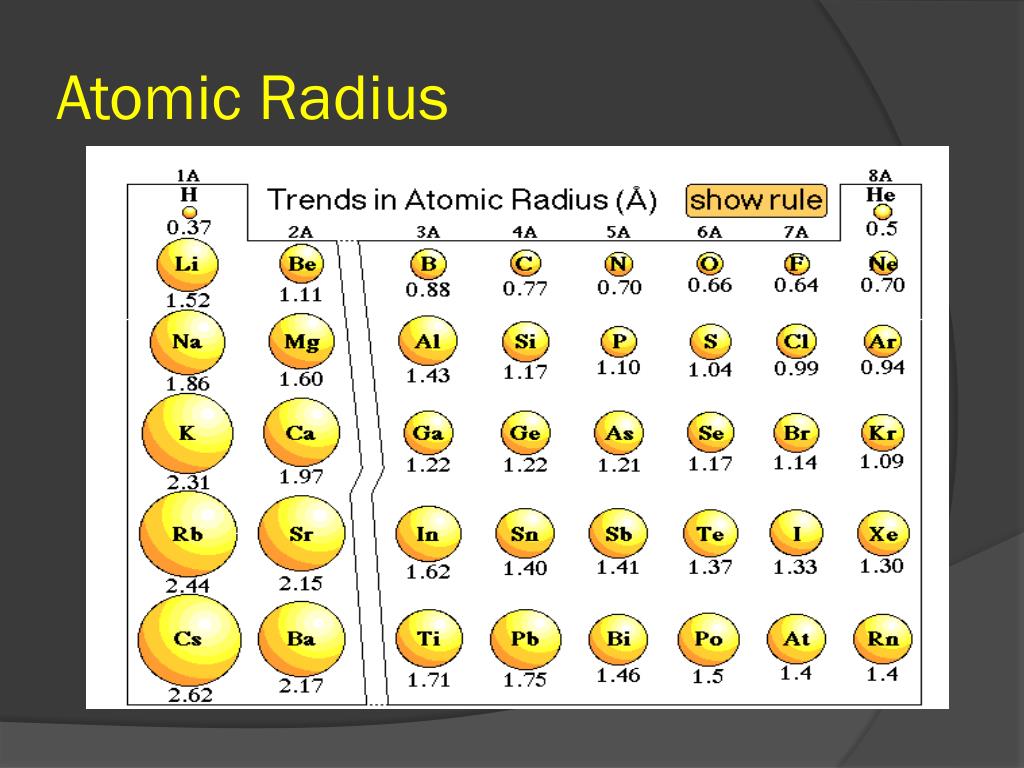
Go over these arguments in your head and try to put the pieces together yourself from here. ionic radius is calculated by measuring the distance between cation and anion. The number of electrons in cation is less than its atom. The atomic and ionic radius of a neutral atom is the same, but many elements exist as anions or cations. For metallic elements, the ionic radius is larger than its atomic radius For metallic elements, the ionic radius is smaller than its atomic radius For non-. Ionic radius is the size of an ion (cation or anion). Compare Ionic Radius with Atomic Radius When electron is removed from an atom cation is formed. We expect POSITIVE ions to be SMALLER than the corresponding atom because there are fewer electrons than protons => an excess of POSITIVE charge => the electron cloud is more attracted to the nucleus on average and becomes more 'concentrated' => $\ce$ carries no unbalanced charge and has comparable valence principal quantum number to the ions, so we expect its radius to fall somewhere between the positive ions (which are smaller) and the negative ions (which are larger). The values range from 30 pm to more than 200 pm.A periodic table coupled with simple electrostatic arguments should suffice to provide at least a rough ordering of ions and atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
